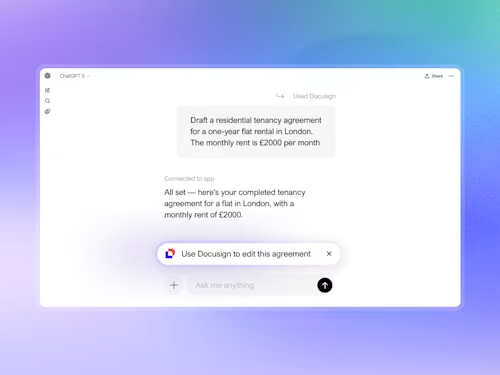
We’re excited to announce Docusign’s partnership with Anthropic to bring Intelligent Agreement Management (IAM) into Claude Cowork. By connecting to Docusign in Cowork, businesses can securely create, review, send, and manage agreements from start to finish — all through natural language prompts in Claude.